Profusa Pivots to Data-as-a-Service Model as 9-Month Hydrogel CGM Disrupts Incumbent 14-Day Lifespans
Date : 2026-04-23
Reading : 95
Profusa’s FY2025 audit reveals a clinical-stage med-tech enterprise bleeding $1.52 million monthly in operating cash while positioning its proprietary Lumee biosensor platforms for a high-stakes 2026 European launch. Operating from two California manufacturing hubs with zero commercial revenue, Profusa aims to disrupt the $2.8 billion continuous glucose monitoring (CGM) market by replacing legacy transcutaneous electronics with a 9-month tissue-integrating hydrogel. With an independent auditor’s going-concern warning and a mathematically depleted organic runway, the company’s survival relies entirely on executing an aggressive reverse recapitalization, securing FDA IDE/PMA authorizations, and mitigating single-source bottlenecks before capital markets tighten.
Financial Health & Capital Structure: The Cost of a SPAC Transition
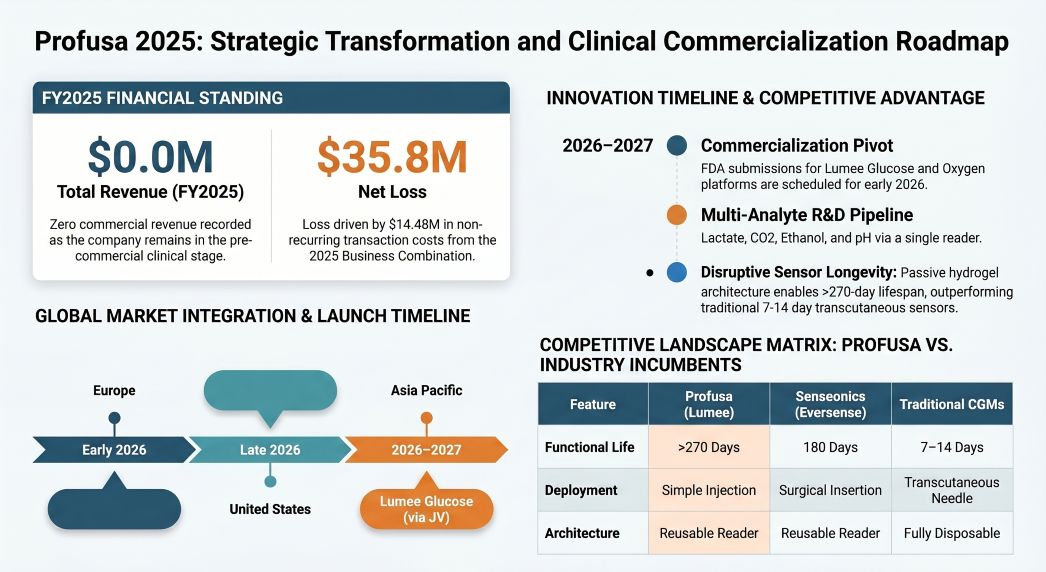
Profusa’s FY2025 financial disclosures reflect the severe capital friction inherent in bringing Class III medical devices to market via a reverse recapitalization. Total revenue flatlined at $0 (down from a $100,000 historical baseline of NIH/DARPA grants in 2024), yielding mathematically undefined gross and EBITDA margins.
The fiscal architecture is dominated by systemic financing challenges rather than core operating expenditures. Net loss widened by 288.1% to $(35.82 million), driven heavily by $14.48 million in direct Business Combination transaction costs and $2.57 million in ELOC warrant issuance fees. Standard clinical-stage cash burn—with R&D rising 74.4% to $2.8 million to support Contract Research Organization (CRO) milestones—was dwarfed by corporate financial engineering.
Most notably, management executed an unorthodox treasury maneuver for a pre-revenue med-tech firm, utilizing $2.0 million of critical liquidity to purchase 16.51 units of Bitcoin. Accounting under ASU 2023-08 forced a non-operating impairment loss of $0.56 million, exacerbating an already fragile balance sheet before the strategy was terminated in Q1 2026. Closing FY2025 with just $1.78 million in cash against a $14.5 million near-term capital requirement through December 2026, Profusa is mathematically dependent on dilutive PIPE convertible notes and an Equity Line of Credit (ELOC) from Ascent Partners Fund LLC to avoid insolvency or Nasdaq delisting.
Figure Profusa 2025 Strategic Transformation and Clinical Commercialization Roadmap
 Operational Moats & The DaaS Pivot
Operational Moats & The DaaS Pivot
Despite immense capital structure stress, Profusa maintains a structurally disruptive operational moat. The continuous glucose monitoring (CGM) sector—dominated by Dexcom (G6), Abbott (Freestyle Libre 2), and Medtronic—relies on transdermal sensors that trigger an immune "foreign body response," limiting functional lifespans to 7–14 days.
Profusa bypasses this biological barrier. The Lumee platform deploys a 400-micron passive hydrogel via a simple hypodermic injection, integrating with subcutaneous tissue to achieve a >270-day (9-month) lifespan while maintaining an 11.7% Mean Absolute Relative Difference (MARD). Crucially, this decoupled hardware architecture—a cheap consumable sensor paired with a reusable optical reader—lowers the economic barrier to entry, expanding the Total Addressable Market (TAM) beyond brittle Type 1 diabetics into the vastly larger non-insulin-dependent Type 2 and pre-diabetic demographics.
To monetize this, Profusa is executing a strategic pivot from pure hardware sales to a recurring Data-as-a-Service (DaaS) model. By commoditizing the sensor deployment—targeting direct-to-pharmacy B2C channels for Lumee Glucose and direct-to-hospital B2B sales for Lumee Oxygen—the firm plans to aggregate real-time biochemical data streams. This shifts their valuation framework toward high-margin data services, powering third-party AI telemedicine platforms without bearing prohibitive direct-to-consumer acquisition costs.
Supply Chain Asymmetry: High Vertical Integration Masking Upstream Vulnerability
Profusa’s supply chain execution presents a stark dichotomy between downstream manufacturing control and extreme upstream vulnerability. The firm exhibits a high degree of vertical integration, bringing >90% of the implantable sensor production inside its California-based ISO-compliant facilities utilizing Class 7 controlled environment rooms (CER). This insulates Profusa from typical Contract Manufacturing Organization (CMO) quality fade and protects proprietary hydrogel trade secrets.
However, this internal capacity masks severe single-source bottlenecks. The foundational biochemical components of the Lumee platform rely on 100% sole-source vendor dependencies:
Hydrogel Microbeads: Microbeads AS
Glucose Sensing Dye: Laurus Labs
Reference Dye: Biotium
Bacterial Catalase: Roche
Any disruption from these vendors—or macroeconomic friction stemming from global logistics chokepoints—would require extensive product redesign and new FDA 510(k)/PMA supplements, immediately halting commercial supply. Furthermore, the geographic concentration of Profusa's unhedged California facilities (which explicitly lack earthquake insurance) leaves production acutely exposed to climate and seismic events.
HDIN Institutional Perspective: The 2030 Patent Cliff and Regulatory Binary
Our strategic audit of Profusa highlights a high-beta binary setup. The technology’s superiority over Senseonics' Eversense E3 (which requires invasive surgical insertion and only lasts 180 days) is clinically evident, but commercialization is operating on a compressed timeline.
The firm faces an impending foundational "patent cliff" in 2030–2031, when the core patents for the "Tissue-Integrating Sensors" expire across the U.S., Canada, and Europe. While analyte-specific chemistries remain protected through 2040, the expiration of the base hydrogel scaffolding opens the door to generic competitors utilizing the same injectable framework. Compounding this risk are U.S. government "march-in" rights tied to legacy DARPA and NIH funding, theoretically permitting forced licensing if national health concerns dictate.
Coupled with impending Medicare sequestration cuts placing downward pressure on Category I CPT code reimbursements and rising compliance costs linked to GDPR and CCPA data privacy frameworks, Profusa has no margin for error. Executive compensation—currently misaligned and heavily skewed toward SPAC completion bonuses rather than long-term FDA clinical endpoints—must be aggressively restructured. Management must lock in EU CE Mark MDR migration and execute the U.S. late-2026 launch before the 2030 IP cliff severely compresses their platform valuation.
Presentation Download & Video Access
Presentation Download: Click the PDF download link under 'Related Topics' to access the presentation of this report.
Video Link: Click this link to watch the YouTube video.
About HDIN Research
HDIN Research is a premier global market intelligence and strategic advisory firm specializing in institutional-grade financial analysis, supply chain audits, and macroeconomic forecasting. Our dedicated sector analysts deliver actionable, data-driven insights tailored for private equity, hedge funds, and corporate strategy teams. Visit us at www.hdinresearch.com.
"This intelligence report was authored by HDIN Research analysts following a rigorous audit of official corporate filings. AI was utilized for data synthesis and structural drafting, with all strategic insights and financial data verified by our editorial board to ensure professional accuracy and compliance with 2026 search standards."
Financial Health & Capital Structure: The Cost of a SPAC Transition
Profusa’s FY2025 financial disclosures reflect the severe capital friction inherent in bringing Class III medical devices to market via a reverse recapitalization. Total revenue flatlined at $0 (down from a $100,000 historical baseline of NIH/DARPA grants in 2024), yielding mathematically undefined gross and EBITDA margins.
The fiscal architecture is dominated by systemic financing challenges rather than core operating expenditures. Net loss widened by 288.1% to $(35.82 million), driven heavily by $14.48 million in direct Business Combination transaction costs and $2.57 million in ELOC warrant issuance fees. Standard clinical-stage cash burn—with R&D rising 74.4% to $2.8 million to support Contract Research Organization (CRO) milestones—was dwarfed by corporate financial engineering.
Most notably, management executed an unorthodox treasury maneuver for a pre-revenue med-tech firm, utilizing $2.0 million of critical liquidity to purchase 16.51 units of Bitcoin. Accounting under ASU 2023-08 forced a non-operating impairment loss of $0.56 million, exacerbating an already fragile balance sheet before the strategy was terminated in Q1 2026. Closing FY2025 with just $1.78 million in cash against a $14.5 million near-term capital requirement through December 2026, Profusa is mathematically dependent on dilutive PIPE convertible notes and an Equity Line of Credit (ELOC) from Ascent Partners Fund LLC to avoid insolvency or Nasdaq delisting.
Figure Profusa 2025 Strategic Transformation and Clinical Commercialization Roadmap
 Operational Moats & The DaaS Pivot
Operational Moats & The DaaS PivotDespite immense capital structure stress, Profusa maintains a structurally disruptive operational moat. The continuous glucose monitoring (CGM) sector—dominated by Dexcom (G6), Abbott (Freestyle Libre 2), and Medtronic—relies on transdermal sensors that trigger an immune "foreign body response," limiting functional lifespans to 7–14 days.
Profusa bypasses this biological barrier. The Lumee platform deploys a 400-micron passive hydrogel via a simple hypodermic injection, integrating with subcutaneous tissue to achieve a >270-day (9-month) lifespan while maintaining an 11.7% Mean Absolute Relative Difference (MARD). Crucially, this decoupled hardware architecture—a cheap consumable sensor paired with a reusable optical reader—lowers the economic barrier to entry, expanding the Total Addressable Market (TAM) beyond brittle Type 1 diabetics into the vastly larger non-insulin-dependent Type 2 and pre-diabetic demographics.
To monetize this, Profusa is executing a strategic pivot from pure hardware sales to a recurring Data-as-a-Service (DaaS) model. By commoditizing the sensor deployment—targeting direct-to-pharmacy B2C channels for Lumee Glucose and direct-to-hospital B2B sales for Lumee Oxygen—the firm plans to aggregate real-time biochemical data streams. This shifts their valuation framework toward high-margin data services, powering third-party AI telemedicine platforms without bearing prohibitive direct-to-consumer acquisition costs.
Supply Chain Asymmetry: High Vertical Integration Masking Upstream Vulnerability
Profusa’s supply chain execution presents a stark dichotomy between downstream manufacturing control and extreme upstream vulnerability. The firm exhibits a high degree of vertical integration, bringing >90% of the implantable sensor production inside its California-based ISO-compliant facilities utilizing Class 7 controlled environment rooms (CER). This insulates Profusa from typical Contract Manufacturing Organization (CMO) quality fade and protects proprietary hydrogel trade secrets.
However, this internal capacity masks severe single-source bottlenecks. The foundational biochemical components of the Lumee platform rely on 100% sole-source vendor dependencies:
Hydrogel Microbeads: Microbeads AS
Glucose Sensing Dye: Laurus Labs
Reference Dye: Biotium
Bacterial Catalase: Roche
Any disruption from these vendors—or macroeconomic friction stemming from global logistics chokepoints—would require extensive product redesign and new FDA 510(k)/PMA supplements, immediately halting commercial supply. Furthermore, the geographic concentration of Profusa's unhedged California facilities (which explicitly lack earthquake insurance) leaves production acutely exposed to climate and seismic events.
HDIN Institutional Perspective: The 2030 Patent Cliff and Regulatory Binary
Our strategic audit of Profusa highlights a high-beta binary setup. The technology’s superiority over Senseonics' Eversense E3 (which requires invasive surgical insertion and only lasts 180 days) is clinically evident, but commercialization is operating on a compressed timeline.
The firm faces an impending foundational "patent cliff" in 2030–2031, when the core patents for the "Tissue-Integrating Sensors" expire across the U.S., Canada, and Europe. While analyte-specific chemistries remain protected through 2040, the expiration of the base hydrogel scaffolding opens the door to generic competitors utilizing the same injectable framework. Compounding this risk are U.S. government "march-in" rights tied to legacy DARPA and NIH funding, theoretically permitting forced licensing if national health concerns dictate.
Coupled with impending Medicare sequestration cuts placing downward pressure on Category I CPT code reimbursements and rising compliance costs linked to GDPR and CCPA data privacy frameworks, Profusa has no margin for error. Executive compensation—currently misaligned and heavily skewed toward SPAC completion bonuses rather than long-term FDA clinical endpoints—must be aggressively restructured. Management must lock in EU CE Mark MDR migration and execute the U.S. late-2026 launch before the 2030 IP cliff severely compresses their platform valuation.
Presentation Download & Video Access
Presentation Download: Click the PDF download link under 'Related Topics' to access the presentation of this report.
Video Link: Click this link to watch the YouTube video.
About HDIN Research
HDIN Research is a premier global market intelligence and strategic advisory firm specializing in institutional-grade financial analysis, supply chain audits, and macroeconomic forecasting. Our dedicated sector analysts deliver actionable, data-driven insights tailored for private equity, hedge funds, and corporate strategy teams. Visit us at www.hdinresearch.com.
"This intelligence report was authored by HDIN Research analysts following a rigorous audit of official corporate filings. AI was utilized for data synthesis and structural drafting, with all strategic insights and financial data verified by our editorial board to ensure professional accuracy and compliance with 2026 search standards."