Nu-Med Plus Suspends Nitric Oxide Pipeline Amid Capital Depletion as $1.25M Solvency Gap Threatens FY2026 Viability
Date : 2026-04-25
Reading : 89
Nu-Med Plus, Inc. (Who) has completely suspended its nitric oxide (NO) medical device development pipeline (What) following its FY2025 audit (When), retreating to corporate offices in Port Jefferson, NY, and halting operations at its West Jordan, UT R&D facility (Where). The operational paralysis stems from acute insolvency—evidenced by a depleted cash balance of just $661 against $279,189 in current liabilities—forcing independent auditors to issue a severe going concern modification as the company actively seeks a strategic merger to avoid terminal liquidation (Why).
Financial Health & Operational Moats: The Solvency Crisis
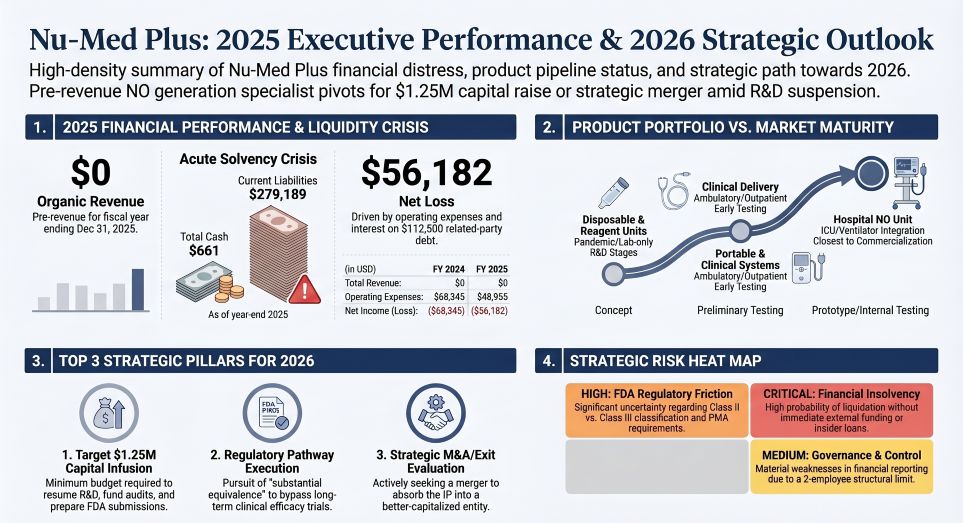
Nu-Med Plus is navigating a critical liquidity vacuum, ending FY2025 with $0 in revenue, total assets of a mere $7,911, and an accumulated deficit exceeding $9.9 million. The company’s financial moat is non-existent, leaving it entirely exposed to predatory financing structures.
The balance sheet is heavily weighted with insider debt and highly dilutive instruments. Specifically, a $100,000 convertible promissory note issued to Your Space, Inc. carries a 5% interest rate that balloons to a punitive 15% penalty upon default in July 2026. Worse for prospective equity holders, the note's conversion mechanism triggers a 20% discount to the share price established during a major exchange listing. With commercial operations halted, the enterprise lacks any organic liquidity, precluding standard inventory de-stocking to generate emergency cash. Management estimates a $1.25 million capital injection is immediately required to fund FY2026 FDA 510(k) preparation and patent maintenance. Without securing an accretive acquisition or a distressed merger, the organic financial runway is strictly zero.
Figure Nu-Med Plus 2025 Executive Performance & 2026 Strategic Outlook
 Supply Chain Paralysis and Entrenched Monopolies
Supply Chain Paralysis and Entrenched Monopolies
Nu-Med's core technological proposition—a proprietary, on-demand NO generation formulation—was designed to dismantle the legacy "razor-and-blade" cylinder model dominated by Mallinckrodt Inc.'s INOMAX system. However, the operational reality reflects total supply chain dormancy.
Because Nu-Med cannot fund third-party clinical sites, Contract Research Organizations (CROs), or external procurement, its manufacturing base remains strictly theoretical. The company cannot activate its supply network to complete prototypes for its Hospital NO Unit or Portable Delivery System. Furthermore, giants like Mallinckrodt utilize deep vertical integration to protect their pressurized tank monopolies and navigate complex environmental handling laws. Nu-Med, lacking even basic environmental liability insurance for toxic tort claims, cannot legally or financially compete in this arena. Since the firm has zero commercial installations, the theoretical margin compression it hoped to force upon legacy tank providers remains an untested whiteboard concept.
HDIN Institutional Perspective: The R&D Capital Trap
From a macroeconomic standpoint, Nu-Med Plus illustrates the fatal vulnerability of micro-cap medtech firms attempting to navigate U.S. regulatory friction without institutional backing. Management's forward-looking strategy hinges on bypassing standard Phase III clinical efficacy trials via the FDA’s 510(k) "substantial equivalence" pathway. Yet, the absence of initial FDA clearance entirely precludes the implementation of cost-pass-through mechanisms to third-party payers or Group Purchasing Organizations (GPOs).
Furthermore, the enterprise is structurally barred from institutional investment due to severe corporate governance deficiencies. Operating with just two employees, zero independent board directors, and declared material weaknesses in internal financial controls by auditor Fruci & Associates II, PLLC, Nu-Med exhibits a distressed agency profile. Insider officers operate simultaneously as fiduciaries and primary creditors ($112,500 in related-party notes). In the event of liquidation, these executive debt claims will legally supersede those of minority equity holders. Nu-Med’s trajectory is a textbook indicator that proprietary intellectual property, absent an aggressive capitalization strategy, rapidly degrades into a stranded asset.
Presentation Download & Video Access
Presentation Download: Click the PDF download link under 'Related Topics' to access the presentation of this report.
Video Link: Click this link to watch the YouTube video.
About HDIN Research:
HDIN Research is a premier provider of institutional-grade market intelligence, forensic financial analysis, and strategic sector forecasting. We specialize in deciphering complex regulatory filings and macroeconomic cycles to deliver actionable insights for hedge funds, private equity, and sovereign wealth portfolios. (www.hdinresearch.com)
This intelligence report was authored by HDIN Research analysts following a rigorous audit of official corporate filings. AI was utilized for data synthesis and structural drafting, with all strategic insights and financial data verified by our editorial board to ensure professional accuracy and compliance with 2026 search standards.
Financial Health & Operational Moats: The Solvency Crisis
Nu-Med Plus is navigating a critical liquidity vacuum, ending FY2025 with $0 in revenue, total assets of a mere $7,911, and an accumulated deficit exceeding $9.9 million. The company’s financial moat is non-existent, leaving it entirely exposed to predatory financing structures.
The balance sheet is heavily weighted with insider debt and highly dilutive instruments. Specifically, a $100,000 convertible promissory note issued to Your Space, Inc. carries a 5% interest rate that balloons to a punitive 15% penalty upon default in July 2026. Worse for prospective equity holders, the note's conversion mechanism triggers a 20% discount to the share price established during a major exchange listing. With commercial operations halted, the enterprise lacks any organic liquidity, precluding standard inventory de-stocking to generate emergency cash. Management estimates a $1.25 million capital injection is immediately required to fund FY2026 FDA 510(k) preparation and patent maintenance. Without securing an accretive acquisition or a distressed merger, the organic financial runway is strictly zero.
Figure Nu-Med Plus 2025 Executive Performance & 2026 Strategic Outlook
 Supply Chain Paralysis and Entrenched Monopolies
Supply Chain Paralysis and Entrenched Monopolies Nu-Med's core technological proposition—a proprietary, on-demand NO generation formulation—was designed to dismantle the legacy "razor-and-blade" cylinder model dominated by Mallinckrodt Inc.'s INOMAX system. However, the operational reality reflects total supply chain dormancy.
Because Nu-Med cannot fund third-party clinical sites, Contract Research Organizations (CROs), or external procurement, its manufacturing base remains strictly theoretical. The company cannot activate its supply network to complete prototypes for its Hospital NO Unit or Portable Delivery System. Furthermore, giants like Mallinckrodt utilize deep vertical integration to protect their pressurized tank monopolies and navigate complex environmental handling laws. Nu-Med, lacking even basic environmental liability insurance for toxic tort claims, cannot legally or financially compete in this arena. Since the firm has zero commercial installations, the theoretical margin compression it hoped to force upon legacy tank providers remains an untested whiteboard concept.
HDIN Institutional Perspective: The R&D Capital Trap
From a macroeconomic standpoint, Nu-Med Plus illustrates the fatal vulnerability of micro-cap medtech firms attempting to navigate U.S. regulatory friction without institutional backing. Management's forward-looking strategy hinges on bypassing standard Phase III clinical efficacy trials via the FDA’s 510(k) "substantial equivalence" pathway. Yet, the absence of initial FDA clearance entirely precludes the implementation of cost-pass-through mechanisms to third-party payers or Group Purchasing Organizations (GPOs).
Furthermore, the enterprise is structurally barred from institutional investment due to severe corporate governance deficiencies. Operating with just two employees, zero independent board directors, and declared material weaknesses in internal financial controls by auditor Fruci & Associates II, PLLC, Nu-Med exhibits a distressed agency profile. Insider officers operate simultaneously as fiduciaries and primary creditors ($112,500 in related-party notes). In the event of liquidation, these executive debt claims will legally supersede those of minority equity holders. Nu-Med’s trajectory is a textbook indicator that proprietary intellectual property, absent an aggressive capitalization strategy, rapidly degrades into a stranded asset.
Presentation Download & Video Access
Presentation Download: Click the PDF download link under 'Related Topics' to access the presentation of this report.
Video Link: Click this link to watch the YouTube video.
About HDIN Research:
HDIN Research is a premier provider of institutional-grade market intelligence, forensic financial analysis, and strategic sector forecasting. We specialize in deciphering complex regulatory filings and macroeconomic cycles to deliver actionable insights for hedge funds, private equity, and sovereign wealth portfolios. (www.hdinresearch.com)
This intelligence report was authored by HDIN Research analysts following a rigorous audit of official corporate filings. AI was utilized for data synthesis and structural drafting, with all strategic insights and financial data verified by our editorial board to ensure professional accuracy and compliance with 2026 search standards.