RetinalGenix Stalls Ophthalmic AI Pivot as FY2025 Audit Reveals $14,774 Cash Runway and 78% R&D Contraction
Date : 2026-04-26
Reading : 55
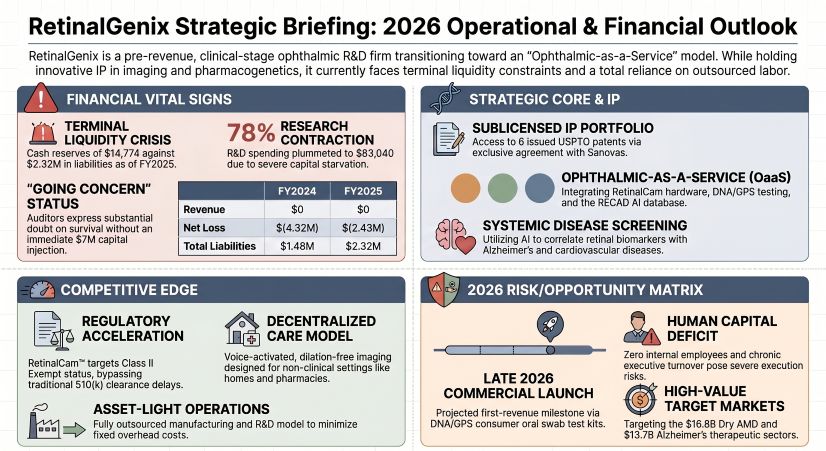
RetinalGenix Technologies Inc. is facing an existential liquidity crisis, holding a dangerously depleted $14,774 in cash against $2.32 million in liabilities following its FY2025 audit. Starved of capital, the California-based pre-revenue med-tech firm slashed R&D spending by 78% in 2025, effectively halting the clinical prototyping of its RetinalCam™ hardware and RECAD™ AI ecosystem. Operations cannot be sustained beyond April 2026 without an immediate $7.0 million capital injection. Consequently, the company remains structurally insolvent, entirely dependent on related-party debt forbearance from principal stockholder Sanovas and highly dilutive micro-cap equity issuances to fund week-to-week survival.
The Illusion of Solvency
A forensic autopsy of RetinalGenix’s FY2025 Form 10-K reveals a corporate shell surviving on life support rather than operational efficiency. While total operating expenses contracted 44% to $2.42 million, this reduction was engineered through capital starvation, not strategic discipline. Research and Development (R&D)—the theoretical moat for any clinical-stage enterprise—plummeted to a mathematically irrelevant $83,040 (representing just 3.4% of total OPEX).
By prioritizing administrative survival over clinical execution, the company has paralyzed its own intellectual property engine. The IP portfolio remains entirely static, reliant on an exclusive worldwide sublicense with related-party Sanovas Ophthalmology LLC. There were zero new patents granted or filed in 2025. Furthermore, the company’s cost structure is heavily distorted by related-party allocations; $661,500 of the $1.30 million G&A expense was funnelled directly to Sanovas. Absent accretive acquisitions or immediate institutional financing, the company's valuation is completely decoupled from its fundamental viability. To offset its $558,350 operating burn, RetinalGenix resorted to selling 232,444 shares at $2.25, further illustrating a reliance on highly dilutive micro-cap equity issuances rather than sustainable debt facilities.
Figure RetinalGenix Strategic Briefing: 2026 0perational & Financial Outlook
 The Asset-Light Fallacy
The Asset-Light Fallacy
RetinalGenix operates a zero-headcount, 100% outsourced model. The absence of vertical integration places the company’s strategic roadmap squarely at the mercy of third-party contract manufacturing organizations (CMOs) like ADM Tronics and unnamed optical engineering consultants.
While broader med-tech incumbents navigate cyclical inventory de-stocking, RetinalGenix carries $0 in inventory (0.0x turnover), meaning it has absolutely no buffer against macroeconomic supply shocks. The company’s 10-K explicitly flags severe single-point-of-failure risks regarding high-resolution imaging sensors sourced from foreign vendors. With zero cost-pass-through mechanisms to absorb supplier inflation or currency volatility, and lead times already stretching beyond three months, the supply chain is highly fragile. Future third-party distributor reliance threatens severe gross margin compression, ensuring that even if the DNA/GPS swab tests reach the market in late 2026, value capture will be aggressively heavily weighted toward external partners.
HDIN Institutional Perspective: Regulatory Vulnerability in the OaaS Transition
RetinalGenix is attempting to orchestrate a sophisticated transition from episodic hardware sales to an "Ophthalmic-as-a-Service" (OaaS) data monopoly via its RECAD™ AI system. However, this strategic pivot is fundamentally compromised by a contradictory regulatory posture.
Management intends to bypass traditional FDA 510(k) clearances by classifying the RetinalCam™ as a Class II exempt device. While this theoretical shortcut accelerates time-to-market and reduces clinical trial expenditures, it inadvertently strips the company of regulatory exclusivity. By lowering the barrier to entry for themselves, they eliminate the regulatory moat against well-capitalized incumbents.
In the broader context of the med-tech macro cycle, RetinalGenix represents the exhaustion of the zero-interest-rate-phenomenon (ZIRP) biotech model. A company entirely devoid of independent board oversight—functioning as a vessel for related-party compensation while farming out critical wet AMD trials to Pearl IRB—cannot realistically disrupt a $16.8 billion dry AMD market. The impending clinical data readout in H1 2027 is a mirage unless the underlying $7.0 million funding chasm is immediately bridged.
Presentation Download & Video Access
Click the PDF download link under 'Related Topics' to access the presentation of this report.
Click this link to watch the YouTube video.
About HDIN Research:
HDIN Research (www.hdinresearch.com) is a premier provider of forensic financial intelligence and institutional equity research, delivering uncompromising, data-driven insights into micro-cap volatility, supply chain vulnerabilities, and macroeconomic market shifts.
*This intelligence report was authored by HDIN Research analysts following a rigorous audit of official corporate filings. AI was utilized for data synthesis and structural drafting, with all strategic insights and financial data verified by our editorial board to ensure professional accuracy and compliance with 2026 search standards.*
The Illusion of Solvency
A forensic autopsy of RetinalGenix’s FY2025 Form 10-K reveals a corporate shell surviving on life support rather than operational efficiency. While total operating expenses contracted 44% to $2.42 million, this reduction was engineered through capital starvation, not strategic discipline. Research and Development (R&D)—the theoretical moat for any clinical-stage enterprise—plummeted to a mathematically irrelevant $83,040 (representing just 3.4% of total OPEX).
By prioritizing administrative survival over clinical execution, the company has paralyzed its own intellectual property engine. The IP portfolio remains entirely static, reliant on an exclusive worldwide sublicense with related-party Sanovas Ophthalmology LLC. There were zero new patents granted or filed in 2025. Furthermore, the company’s cost structure is heavily distorted by related-party allocations; $661,500 of the $1.30 million G&A expense was funnelled directly to Sanovas. Absent accretive acquisitions or immediate institutional financing, the company's valuation is completely decoupled from its fundamental viability. To offset its $558,350 operating burn, RetinalGenix resorted to selling 232,444 shares at $2.25, further illustrating a reliance on highly dilutive micro-cap equity issuances rather than sustainable debt facilities.
Figure RetinalGenix Strategic Briefing: 2026 0perational & Financial Outlook
 The Asset-Light Fallacy
The Asset-Light FallacyRetinalGenix operates a zero-headcount, 100% outsourced model. The absence of vertical integration places the company’s strategic roadmap squarely at the mercy of third-party contract manufacturing organizations (CMOs) like ADM Tronics and unnamed optical engineering consultants.
While broader med-tech incumbents navigate cyclical inventory de-stocking, RetinalGenix carries $0 in inventory (0.0x turnover), meaning it has absolutely no buffer against macroeconomic supply shocks. The company’s 10-K explicitly flags severe single-point-of-failure risks regarding high-resolution imaging sensors sourced from foreign vendors. With zero cost-pass-through mechanisms to absorb supplier inflation or currency volatility, and lead times already stretching beyond three months, the supply chain is highly fragile. Future third-party distributor reliance threatens severe gross margin compression, ensuring that even if the DNA/GPS swab tests reach the market in late 2026, value capture will be aggressively heavily weighted toward external partners.
HDIN Institutional Perspective: Regulatory Vulnerability in the OaaS Transition
RetinalGenix is attempting to orchestrate a sophisticated transition from episodic hardware sales to an "Ophthalmic-as-a-Service" (OaaS) data monopoly via its RECAD™ AI system. However, this strategic pivot is fundamentally compromised by a contradictory regulatory posture.
Management intends to bypass traditional FDA 510(k) clearances by classifying the RetinalCam™ as a Class II exempt device. While this theoretical shortcut accelerates time-to-market and reduces clinical trial expenditures, it inadvertently strips the company of regulatory exclusivity. By lowering the barrier to entry for themselves, they eliminate the regulatory moat against well-capitalized incumbents.
In the broader context of the med-tech macro cycle, RetinalGenix represents the exhaustion of the zero-interest-rate-phenomenon (ZIRP) biotech model. A company entirely devoid of independent board oversight—functioning as a vessel for related-party compensation while farming out critical wet AMD trials to Pearl IRB—cannot realistically disrupt a $16.8 billion dry AMD market. The impending clinical data readout in H1 2027 is a mirage unless the underlying $7.0 million funding chasm is immediately bridged.
Presentation Download & Video Access
Click the PDF download link under 'Related Topics' to access the presentation of this report.
Click this link to watch the YouTube video.
About HDIN Research:
HDIN Research (www.hdinresearch.com) is a premier provider of forensic financial intelligence and institutional equity research, delivering uncompromising, data-driven insights into micro-cap volatility, supply chain vulnerabilities, and macroeconomic market shifts.
*This intelligence report was authored by HDIN Research analysts following a rigorous audit of official corporate filings. AI was utilized for data synthesis and structural drafting, with all strategic insights and financial data verified by our editorial board to ensure professional accuracy and compliance with 2026 search standards.*